Specifications
- Environmental
-
Environmental Operating temperature - 10–40°C (50–104°F)
Operating humidity - less than 95% relative humidity @ 40°C (104°F)
Operating Altitude - -500–3000 m (-1640–9840 ft)
Charging temperature - 10–35°C (50–95°F)
Storage/transportation temperature - -20–60°C (-4–140°F)
Storage/transportation humidity - less than 90% relative humidity @ 60°C (140°F)
Storage/transportation altitude - -500–13100 m (-1640–43000 ft)
-
- Performance: aFHR
-
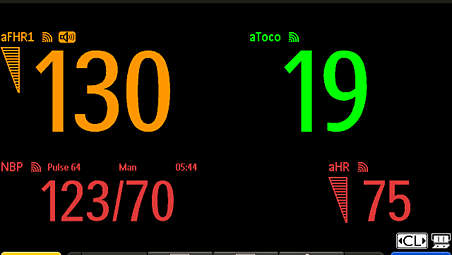
Performance: aFHR Measurement range - 60–240 bpm
Measurement method - Electrocardiography
Resolution display - 1 bpm
Resolution printer - 1/4 bpm
Accuracy - ±1 bpm
-
- Performance: aHR
-
Performance: aHR Measurement method - Electrocardiography
Measurement range - 40–240 bpm
Resolution display - 1 bpm
Resolution printer - 1/4 bpm
Accuracy - ±1 bpm
-
- Performance: aToco
-
Performance: aToco Measurement method - Uterine electromyography
Measurement range - 0–500 μV
Resolution - 0–255 levels representing 100% of the full scale
Accuracy - ±5%
-
- Physical
-
Physical Size (W x H x D) - 63 x 20 x 49 mm (2.5 x 0.8 x 1.9 in)
Weight - 0.07 kg (0.15 lb)
Battery type - Integrated rechargeable Li-Ion battery with battery gauge and cycle counter
Battery runtime (fully charged battery) - min. 16 hours
Battery life - greater than 500 charge-discharge cycles or longer than 4 years
Battery transducer storage time - ≥1 year at 25°C (77°F) (battery should be charged to 40–50% every 6 months)
Battery recharging time - 100% charged ≤3.0 h
Water ingress protection code - IP 67 (immersion up to 1 m water depth for 30 minutes)
Shock resistance - Withstands 1.5 m drop to concrete surface with possible cosmetic damage only
Degree of protection against electrical shock - Type CF
Pod identification - Optical signal element (finder LED)
-
- Environmental
-
Environmental Operating temperature - 10–40°C (50–104°F)
Operating humidity - less than 95% relative humidity @ 40°C (104°F)
-
- Performance: aFHR
-
Performance: aFHR Measurement range - 60–240 bpm
Measurement method - Electrocardiography
-
- Environmental
-
Environmental Operating temperature - 10–40°C (50–104°F)
Operating humidity - less than 95% relative humidity @ 40°C (104°F)
Operating Altitude - -500–3000 m (-1640–9840 ft)
Charging temperature - 10–35°C (50–95°F)
Storage/transportation temperature - -20–60°C (-4–140°F)
Storage/transportation humidity - less than 90% relative humidity @ 60°C (140°F)
Storage/transportation altitude - -500–13100 m (-1640–43000 ft)
-
- Performance: aFHR
-
Performance: aFHR Measurement range - 60–240 bpm
Measurement method - Electrocardiography
Resolution display - 1 bpm
Resolution printer - 1/4 bpm
Accuracy - ±1 bpm
-
- Performance: aHR
-
Performance: aHR Measurement method - Electrocardiography
Measurement range - 40–240 bpm
Resolution display - 1 bpm
Resolution printer - 1/4 bpm
Accuracy - ±1 bpm
-
- Performance: aToco
-
Performance: aToco Measurement method - Uterine electromyography
Measurement range - 0–500 μV
Resolution - 0–255 levels representing 100% of the full scale
Accuracy - ±5%
-
- Physical
-
Physical Size (W x H x D) - 63 x 20 x 49 mm (2.5 x 0.8 x 1.9 in)
Weight - 0.07 kg (0.15 lb)
Battery type - Integrated rechargeable Li-Ion battery with battery gauge and cycle counter
Battery runtime (fully charged battery) - min. 16 hours
Battery life - greater than 500 charge-discharge cycles or longer than 4 years
Battery transducer storage time - ≥1 year at 25°C (77°F) (battery should be charged to 40–50% every 6 months)
Battery recharging time - 100% charged ≤3.0 h
Water ingress protection code - IP 67 (immersion up to 1 m water depth for 30 minutes)
Shock resistance - Withstands 1.5 m drop to concrete surface with possible cosmetic damage only
Degree of protection against electrical shock - Type CF
Pod identification - Optical signal element (finder LED)
-
- ¹ Cohen WR, Hayes-Gill B. . Acta Obstet Gynecol Scand. 2014 Jun ; 93 (6) : 590-5.
- Avalon Beltless Solution has been clinically evaluated in the following studies:
- Fox, D., Coddington, R., Scarf, V. et al. Harnessing technology to enable all women mobility in labour and birth: feasibility of implementing beltless non-invasive fetal ECG applying the NASSS framework. Pilot Feasibility Stud 7, 214 (2021). DOI: 10.1186/s40814-021-00953-6
- Fox, D., Coddington, R., Scarf, V. Wanting to be 'with woman', not with machine: Midwives' experiences of caring for women being continuously monitored in labour. Women and birth 35(4), 387-393. DOI: 10.1016/j.wombi.2021.09.002
- C Reis de Carvalho, P Nogueira, D Ayres-de-Campos. Quality of fetal heart rate monitoring with transabdominal fetal ECG during maternal movement in labor: A prospective study. Acta Obstetricia et Gynecologica Scandinavica 2022. DOI: 10.1111/aogs.14434
- Product may not be available in all geographies, please check with your Philips representative for complete portfolio availability.